Researchers from the University of California, San Diego and UCLA, Soochow University and Westlake University in China, and Marmara University in Turkey, have examined the surface defect-deactivation mechanism in perovskite solar cells using molecules found in tea, coffee and chocolate.
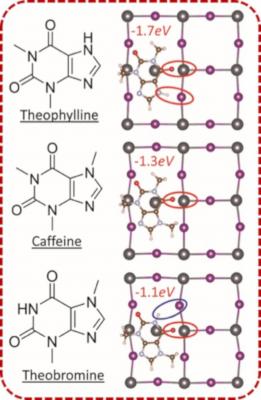
The collaborative team set out to delineate the molecular arrangements that constructively deactivate the surface defects in perovskite solar-cells. Highly-efficient metal-halide perovskite solar cells to date consist of polycrystalline perovskite film that often contains a high density of defects on the surface. These imperfections are the points for charge recombination, which is a major limiting factor in power conversion efficiency (PCE) and stability of perovskite solar cells. However, due to the ionic nature of the perovskite lattice, these defects can be passivated by surface treatment of perovskite with a small molecule.
The team showed that the surface treatment of perovskite using theophylline significantly increased the efficiency over that observed without theophylline. They then showed that to maximize the binding to surface-defects of perovskite, the amine and carbonyl functional groups have to be in an optimal configuration within the xanthine molecule relative to the surface of perovskite.

