Researchers at the Eindhoven University of Technology in the Netherlands have found a way to address the issue of stability in perovskite solar cells by adding a small amount of fluoride during the production process, which was found to increase the stability of such cells.
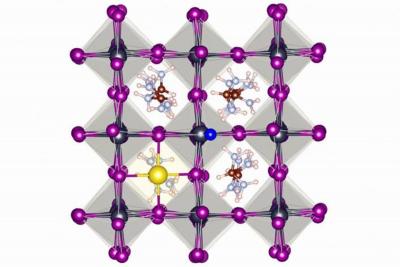 Fluoride stablizes perovskite solar cells by encouraging the formation of strong hydrogen bonds and ionic bonds on the surface of the perovskite material.
Fluoride stablizes perovskite solar cells by encouraging the formation of strong hydrogen bonds and ionic bonds on the surface of the perovskite material.
The scientists found the fluoride ions form a protective layer around perovskite crystals, preventing the ill effects of light, heat and moisture. "Our work has improved the stability of perovskite solar cells considerably," said Shuxia Tao, assistant professor at Eindhoven University of Technology's Center for Computational Energy Research. "Our cells maintain 90% of their efficiency after 1,000 hours under extreme light and heat conditions. This is many times as long as traditional perovskite compounds. We achieve an efficiency of 21.3%, which is a very good starting point for further efficiency gains."
The team plans to continue tweaking the process for adding fluoride to perovskite solar cells. Their goal is to create fluoride-enhanced perovskite solar cells that maintain 85% of their original efficiency after ten to fifteen years.
"We expect it will take another five to ten years for these cells to become a commercially viable product. Not only do we need to further improve their efficiency and stability, we also need to gain a better theoretical understanding of the relevant mechanisms at the atomic scale," said Tao. "We still don't have all the answers to why some materials are more effective than others in increasing the long-term stability of these cells."