A team of scientists at EPFL has come up with an efficient solution to the lead problem of perovskite solar cells, which involves using a transparent phosphate salt that does not block solar light and hence doesn't affect performance.
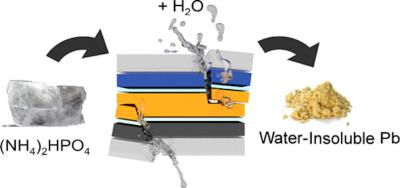
In case the solar panel fails, the phosphate salt immediately reacts with lead to produce a water-insoluble compound that cannot leach out to the soil, and which can be recycled.
The problem is that in most of the halide perovskites lead can dissolve in water. This water solubility and solubility in other solvents is actually a great advantage, as it makes building perovskite solar panels simpler and inexpensive - another advantage along with their performance. But the water solubility of lead can become a real environmental and health hazard when the panel breaks or gets wet, e.g. when it rains.
The lead must be captured before it gets to the soil, and it must be possible to recycle it. This issue has drawn much and intensive research because it is the main obstacle for regulatory authorities approving the production of perovskite solar cells on a large, commercial scale. However, attempts to synthesize non-water-soluble and lead-free perovskites have thus far yielded poor performance.
"A few years ago, we discovered that cheap and transparent phosphate salt crystals, like those in soil fertilizers, can be incorporated into various parts of the sandwich-like lead halide perovskite devices, like photodetectors, LEDs or solar cells," says Endre Horváth, the study's first author. "These salts instantaneously react with lead ions in the presence of water, and precipitate them into extremely non-water-soluble lead phosphates."
"The 'fail-safe' chemistry keeps lead ions from leaching out and can render perovskite devices safer to use in the environment or close to humans," says Márton Kollár, the chemist behind the growth of perovskite crystals.
"We show that this approach can be used to build functional photodetectors, and we suggest that the broad community of researchers and R&D centers working on various devices like solar cells and light-emitting diodes implements it in their respective prototypes," adds Pavao AndriÄevic, who characterized the sensitive photodetectors.
Forró concludes: "This is an extremely important study - I would say, a central one - for large-scale commercialization of perovskite-based solar cells."