A collaboration between KAUST and Oxford University researchers has designed a strategy that grows perovskites into centimeter-scale, highly pure crystals thanks to the effect of surface tension. In their natural state, perovskites have difficultly moving solar-generated electricity because they crystallize with randomly oriented grains. The team is working on ways to speed up the flow of these charge carriers using inverse temperature crystallization (ITC) - a technique that uses special organic liquids and thermal energy to force perovskites to solidify into structures resembling single crystals (the optimal arrangements for device purposes).
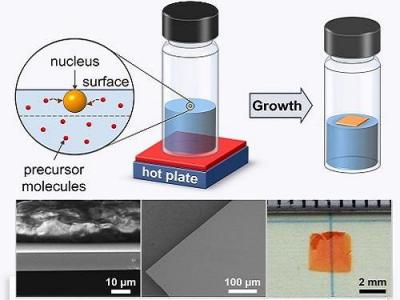
While ITC produces high-quality perovskites far faster than conventional chemical methods, the intriguing mechanisms that initiate crystallization in hot organic liquids are poorly understood. The team recalls spotting a key piece of evidence during efforts to adapt ITC toward large-scale manufacturing. "At some point, we realized that when crystals appeared, it was usually at the solution's surface," the researchers said. "And this was particularly true when we used concentrated solutions".
Interestingly, the researchers found that complexes located at the solution surface can experience additional forces due to surface tension. The extra pull provided by the surface makes it much easier to separate the solvent-perovskite complexes and nucleate crystals that float on top of the liquid. This helped the team produce centimeter-sized, ultrathin single crystals and prototype a photodetector with characteristics comparable to state-of-the-art devices. Although the single crystals are currently fragile and difficult to handle due to their microscale thicknesses, the team explains that this method could help direct the perovskite growth onto specific substrates.
"Taking into account the roles of interfaces and surface tension could have a fundamental impact," the scientists say, "we can get large-area growth, and it's not limited to specific metal cations-you could have a library of materials with perovskite structures."

