Researchers at Brown University and University of Nebraska - Lincoln (UNL) have come up with a new titanium-based material for making lead-free, inorganic perovskite solar cells. The team shows that the material has significant potential, especially for making tandem solar cells.
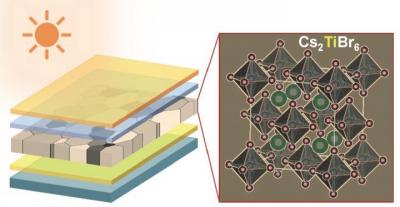
"Titanium is an abundant, robust and biocompatible element that, until now, has been largely overlooked in perovskite research," said the senior author of the new paper. "We showed that it's possible to use titanium-based material to make thin-film perovskites and that the material has favorable properties for solar applications which can be tuned."
The team made semi-transparent perovskite films with a bandgap of 1.8 electron volts, which is considered to be ideal for tandem solar applications. The material had a conversion efficiency of 3.3%, which is well below that of lead-based cells, but a good start for an all-new material, the researchers say.
The researchers used a high-temperature evaporation method to prepare the films, but the team is reportedly investigating alternative methods. "We are also looking for new low-temperature and solvent-based methods to reduce the potential cost of cell fabrication".
The research showed the material has several advantages over other lead-free perovskite alternatives. One contender for a lead-free perovskite is a material made largely from tin, which rusts easily when exposed to the environment. Titanium, on the hand, is rust-resistant. The titanium-perovskite also has an open-circuit voltage - a measure of the total voltage available from a solar cell - of over one volt. Other lead-free perovskites generally produce voltage smaller than 0.6 volts.
"Open-circuit voltage is a key property that we can use to evaluate the potential of a solar cell material," the team said. "So, having such a high value at the outset is very promising."
The researchers say that material's relatively large bandgap compared to silicon makes it a prime candidate to serve as the top layer in a tandem solar cell. The titanium-perovskite upper layer would absorb the higher-energy photons from the sun that the lower silicon layer can't absorb because of its smaller bandgap. Meanwhile, lower energy photons would pass through the semi-transparent upper layer to be absorbed by the silicon, thereby increasing the cell's total absorption capacity.
"Tandem cells are the low-hanging fruit when it comes to perovskites," the scientists said. "We're not looking to replace existing silicon technology just yet, but instead we're looking to boost it. So if you can make a lead-free tandem cell that's stable, then that's a winner. This new material looks like a good candidate."