Researchers at the Los Alamos National Laboratory (LANL) in New Mexico, USA have found out why perovskite solar cells degrade in sunlight, as well as how the devices can 'self-heal' by being given time in the dark. The research, that determined that photo-degradation in perovskite cells is a purely electronic process due to charge accumulation without chemical damage to the crystal structure, could help catalyze the promising solar technology.
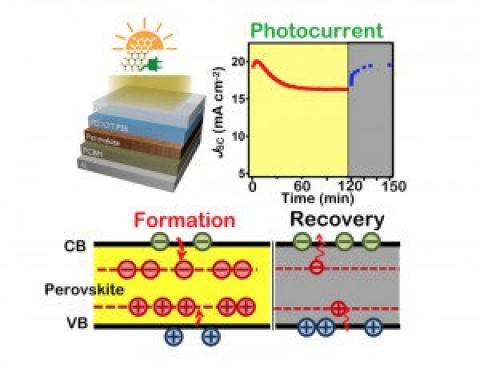
The new finding, that organometallic halide semiconducting perovskite solar cells degrade under continuous sunlight but recover in the dark is seen as very good news as it means the process is of physical nature rather than chemical decomposition. According to the researchers: 'After extensive characterization electronically and optically, we found it was the charge accumulation in the cell that reduces the photo-generated electrical current. Our team predicts the charge accumulation is initiated by the charged trap states, called small polaron states, which is only activated under light.'
Encouraged by this knowledge, the LANL team was able to devise a solution to help mitigate the degradation issue in perovskite solar cells. They found there are two ways to stabilize the cell: one is to reduce the temperature to 0 degree Celsius; the cell can be stable for a longer time in light. The other way is to take advantage of the recovery nature. In the dark, upon heating the cell, the performance can be fully restored within a minute.
The study could have significant impact on the future of perovskite solar cells. The reason for the degradation is two-fold: One aspect is the chemical instability due to environment and the other part of the problem is photo-stability, where light could potentially kill the cell. 'The chemical stability can be circumvented by capping or encapsulation, but the photo instability is not acceptable for commercial use. This study tried to understand the physics behind the photo-degradation, which will shed light to searching for a photo-stable hybrid perovskite material for real applications.

