Research by scientists at the Eindhoven University of Technology and universities in China and the US sheds new light on the causes of the degradation perovskites undergo when exposed to sunlight and paves the way for designing new perovskite compositions for the ultimate stable solar cells.
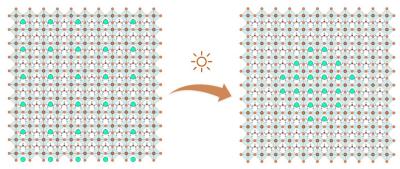
The new research focuses on perovskite solar cells made from formamidinium-caesium lead iodide, a halide compound that has become increasingly popular as it combines high efficiency and reasonable heat resistance with low manufacturing costs.
From this they conclude that sunlight generates charged particles in the perovskite, which tend to flow to places in the solar panel where the band gap (the minimum amount of energy needed for generating the free electrons) is lowest, in this case the formamidinium perovskite. The resulting energy differences make the mixed compounds that worked together so well to make the cell efficient, fall apart into separate clusters.
It appears that especially the caesium-heavy clusters are photoinactive and current-blocking, limiting the performance of the device.
According to Shuxia Tao, who together with PhD candidate Zehua Chen and her colleague Geert Brocks was responsible for the TU/e part of the research, the new findings are one step further to finding the way to possible solutions.
"By combining macroscopic tests, microscopic materials characterization and atomistic modelling, we were able to thoroughly understand the instability of halide perovskites that are intrinsic to device operation. This opens the possibility for designing new perovskite compositions for the ultimate stable solar cells."
Possible strategies include using additives to enhance the chemical interaction inside the materials in the panels, tuning the band gaps by using other elements like bromide and rubidium instead of iodide and caesium, or modifying the energy levels to extract photo-carriers more efficiently.
Tao stresses that more research is needed to see what solution works best. In addition, separation of halide compounds is not the only cause for perovskite degradation. These additional causes require separate analysis.