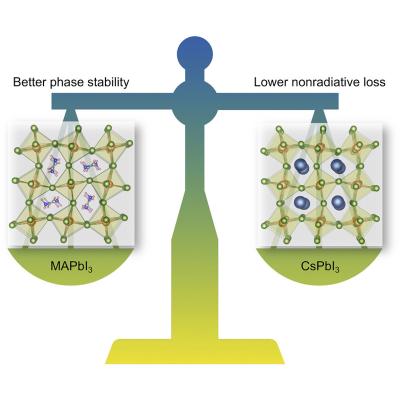
Researchers at UC Santa Barbara recently conducted a research that disproved the common belief that organic molecules are crucial to achieving PSCs' impressive performance because they suppress defect-assisted carrier recombination. Not only was this assumption shown to be incorrect, but the team also found that all-inorganic materials have the potential for outperforming hybrid perovskites.
“To compare the materials, we performed comprehensive simulations of the recombination mechanisms,†explained Xie Zhang, lead researcher on the study. “When light shines on a solar-cell material, the photo-generated carriers generate a current; recombination at defects destroys some of those carriers and hence lowers the efficiency. Defects thus act as efficiency killers.â€
Sorting out these processes experimentally is extremely difficult, but state-of-the-art quantum-mechanical calculations can accurately predict the recombination rates, thanks to new methodology that was developed in the group of UCSB materials professor Chris Van de Walle, who credited Mark Turiansky, a senior graduate student in the group, with helping to write the code to calculate the recombination rates.
“Our methods are very powerful for determining which defects cause carrier loss,†Turiansky said. “It is exciting to see the approach applied to one of the critical issues of our time, namely the efficient generation of renewable energy.â€
Running the simulations showed that defects common to both materials give rise to comparable (and relatively benign) levels of recombination. However, the organic molecule in the hybrid perovskite can break up; when loss of hydrogen atoms occurs, the resulting “vacancies†strongly decrease efficiency. The presence of the molecule is thus a detriment, rather than an asset, to the overall efficiency of the material.
Why, then, has this not been noticed experimentally? Mainly because it is more difficult to grow high-quality layers of the all-inorganic materials. They have a tendency to adopt other crystal structures, and promoting the formation of the desired structure requires greater experimental effort. Recent research has shown, however, that achieving the preferred structure is definitely feasible. Still, the difficulty explains why the all-inorganic perovskites have not received as much attention to date.
“We hope that our findings about the expected efficiency will stimulate more activities directed at producing inorganic perovskites,†concluded Van de Walle.