Researchers from Lomonosov Moscow State University in Russia explained how changing the ratio of components forming the light-absorbing layer of a perovskite solar cell influences the structure of created films and battery efficiency.
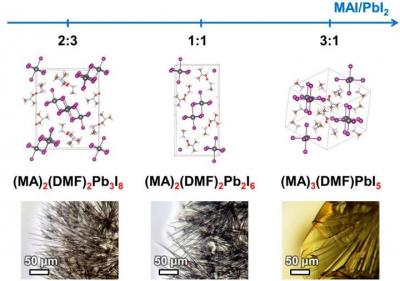
Perovskites can be used to create perovskite solar batteries, which are a relatively new area of R&D, but are showing promise in terms of efficiency and function. In their previous work, the researchers found out that filiform (wire-like) hybrids of perovskites have acquired their shape because of the structure of intermediate compounds, which are formed during the process of perovskite crystallization. The team has discovered a whole group of these compounds, every one of which is a crystalline solvate. The crystalline solvates are crystalline compounds with the molecules of the precursor components' solvent built into their structure. The dissolved components precipitate from the solution and form a crystalline film of perovskite.
Such intermediate compounds are unstable, so the authors used synchrotron radiation and low temperatures to cool the crystals to the temperature of -173 °C. The freezing allowed the scientists to stop the decomposition of the crystals and to run the necessary measurements to become able next to determine the structure of the solvates.
Additionally, the researchers have studied the thermal stability of the obtained compounds and managed to calculate the energy of their formation using quantum-chemical modeling. Knowing the formation energy allows to explain why certain crystals are formed when using different solvents.
The authors have also learned that the ratio of reagents in solution specifically determines which intermediate compound will form in the process of crystallization. Crystal structure of the intermediate compound defines the shape of the perovskite crystals formed, which determines the structure of the light-absorbing layer. This structure, in turn, influences the output of the solar battery created.

