Perovskites are materials that share a crystal structure similar to the mineral called perovskite, which consists of calcium titanium oxide (CaTiO3).
Depending on which atoms/molecules are used in the structure, perovskites can possess an impressive array of interesting properties including superconductivity, ferroelectricity, charge ordering, spin dependent transport and much more. Perovskites therefore hold exciting opportunities for physicists, chemists and material scientists.
Lead-based perovskites have become key materials in photovoltaics research thanks to their facile solution processability and impressive performance. However, despite their great potential, a persistent threat on future use and commercialization is the issue of toxicity and lead-content.
Lead (and its oxide form) is highly hazardous to animals and humans. Hence, succeeding to develop lead-free perovskites that could be used instead of the lead-based ones is a much desired accomplishment. While producing lead-free perovskite materials is not very hard to do, and various candidates exist (Sn-based perovskites, for example) these tend to perform poorly compared to lead-based perovskite materials, and exhibit limited optoelectronic performance and stability. However, vigorous research is taking place and hopefully it will yield lead-free perovskites that can serve as a viable replacement to lead-based ones.
The latest Perovskite toxicity news:
Researchers develop Sn-based perovskite material with a wide visible-light absorption band
Semiconductors that can exploit the omnipresent visible spectrum of light for different technological applications are highly sought after, but such semiconductors are often dexpensive and toxic. A group of scientists from Tokyo Institute of Technology and Kyushu University have collaborated to develop a low-cost and non-toxic narrow-gap semiconductor material with potential 'light-based' or photofunctional applications.
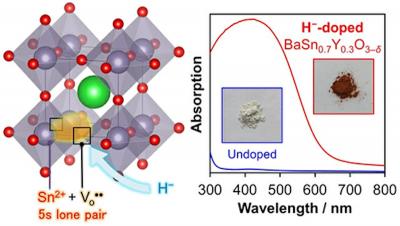
Tin-containing oxide semiconductors are cheaper than most semiconductor materials, but their photofunctional applications are constrained by a wide optical band gap. The team of scientists, led by Dr. Kazuhiko Maeda, Associate Professor at the Department of Chemistry, Tokyo Institute of Technology, developed a perovskite-based semiconductor material that is free of toxic lead and can absorb a wide range of visible light.
Swansea team introduces a nontoxic, biodegradable, green alternative to harmful solvents used to make PSCs
Scientists from Swansea University have found that a non-toxic biodegradable solvent called GVL could remove a major barrier to the production of printed carbon perovskite solar cells.
A known barrier to the large-scale manufacture and commercialization of these cells is the solvents used to control crystallization of the perovskite during fabrication because they are made from toxic, unsustainable materials that are banned in many countries. Now, researchers at SPECIFIC Innovation and Knowledge Centre, Swansea University, have discovered that a non-toxic, biodegradable solvent called GVL (γ-Valerolactone) could replace these solvents without impacting cell performance.
NREL scientists assess health and environmental impacts of solvents for producing perovskite solar cells
Scientists at the National Renewable Energy Laboratory (NREL) have investigated how to manufacture perovskite materials and solar technology with human health in mind.
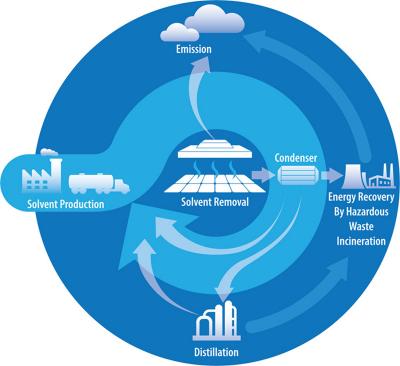
Newly published research points to the safest choices for solvents needed to make perovskite solar cells. Unlike silicon solar panels, which require an industrial process, perovskite solar panels can be made using chemicals and produced using a roll-to-roll printing method or spray coating. The process also costs less and takes less time than manufacturing silicon panels.
Bifacial perovskite solar cells could become more environmentally friendly than conventional crystalline silicon devices
Scientists from the South Dakota School of Mines and Technology and Michigan State, Toledo and Wisconsin universities have found bifacial perovskite PV cells have the potential to become more environmentally friendly than conventional crystalline silicon devices, due to their increased energy yield.
The scientists examined sites at Toledo, in Ohio and Golden, Colorado, to take account of high and low latitude and humidity conditions. The researchers analyzed the environmental impact of single-junction, bifacial perovskite cells with high and low bandgaps, and multi-junction devices of the same type with two-terminal (2T) and four-terminal (4T) structures. They quantified the life cycle energy production from the cells under real-world conditions and benefiting from diverse albedo environments including installation above sandstone, concrete, grass and snow.
Sustainable lead management system could solve toxicity issue and promote commercialization of PSCs
Scientists from the Korea Institute of Machinery and Materials, Kyungpook National University, Sungkyunkwan University, Sejong University and Yonsei University in Korea, in collaboration with Uppsala University in Sweden, Imperial College London and National Renewable Energy Laboratory in the U.S, recently devised a way to sustainably collect pollutants secreted from PSCs without sacrificing the panel itself. Using this new approach, the scientists were able to safely recycle 99.7% of lead in their samples.
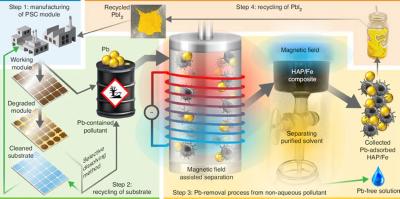
In the study describing their work, the researchers explain that they're not the first to attempt to tackle this issue, but that previous approaches to adsorbing lead have been limited by the number of naturally occurring lead solvents.
Silver-based perovskites with anti-microbial properties could be useful in tissue engineering
A new study, led by Dr. Shayanti Mukherjee at the Australian Hudson Institute (a leading Australian translational medical research institute), has found that perovskites materials can have anti-microbial properties, without toxic side effects to human cells.
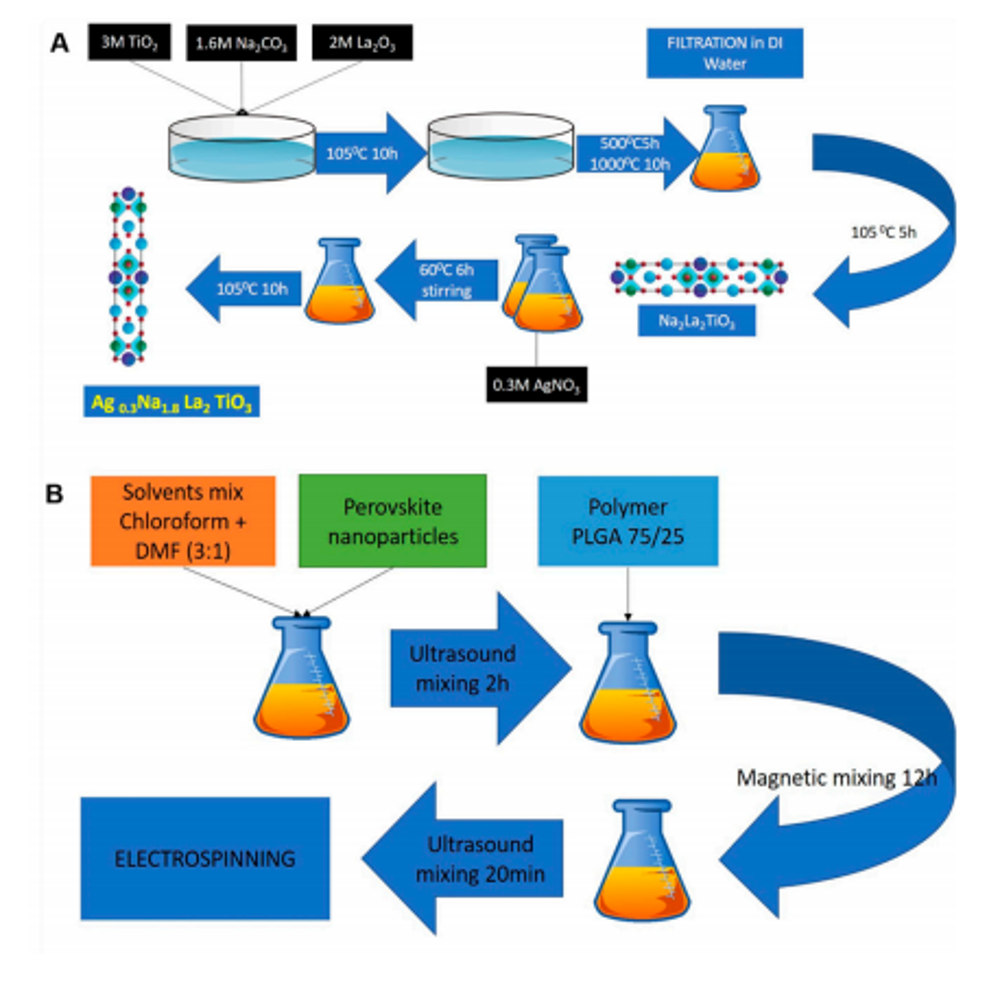 (A) process of perovskite synthesis; (B) preparation of electrospinning solution with perovskite. Image from Nanomaterials
(A) process of perovskite synthesis; (B) preparation of electrospinning solution with perovskite. Image from Nanomaterials
Dr Mukherjee and her team, who already have a significant program researching new bio-degradable nanomeshes to revolutionize treatments for pelvic organ prolapse (POP), have shown for the first time that perovskites can be used as additives to engineer human tissue implants.
Peppermint oil and walnut aroma to enable HT material that prevents lead leakage in perovskite solar cells
Researchers at the Pohang University of Science & Technology (POSTECH) have developed eco-friendly-solvent processable hole transport polymers by using peppermint oil and walnut aroma food additives and the polymer can prevent lead leakage.
The POSTECH research team consisted of Prof. Taiho Park and Junwoo Lee, that developed Alkoxy-PTEG - hole transport polymers that could be dissolved in peppermint oil, by applying ethylene glycol side chains when producing perovskite solar cells. Also, the team confirmed that this polymer captured leaking lead in aging perovskite solar cells.
New lead sequestration technique could make for safer lead-based perovskite solar cells
Researchers at Northern Illinois University and the U.S. Department of Energy's (DOE) National Renewable Energy Laboratory (NREL) in Colorado have reported on a potential breakthrough in the development of hybrid perovskite solar cells.
Led by Tao Xu of NIU and Kai Zhu of NREL, the scientists have developed a technique to sequester the lead used to make perovskite solar cells and minimize potential toxic leakage by applying lead-absorbing films to the front and back of the solar cell.
HZB-led team finds that plants absorb more lead from perovskite solar cells than expected
Researchers led by Prof. Antonio Abate at the Helmholtz-Zentrum Berlin have designed a study to investigate lead hazards relating to perovskite soar cells. They cooperated with plant scientists from the Fujian Agriculture and Forestry University, China, where the experiments were carried out, and with a group from the university of Naples, Italy.
 Mint plants grown on control soil (left) and perovskite-contaminated soil (right). Credit: Nature
Mint plants grown on control soil (left) and perovskite-contaminated soil (right). Credit: Nature
The plant experts prepared contaminated soil samples with different concentrations of lead from either perovskite solar cells or other lead sources and cultivated different plants. After a growth period, they analyzed the lead content in leaves and other parts of the plant. They found that lead from perovskite solar cells is 10 times more bioavailable than lead from other industrial sources.
Chalcogenide perovksites found promising for PV and waste heat recovery
Researchers from Lehigh University in Pennsylvania have found that metal chalcogenide perovskites can be used as a thermoelectric material that can convert thermal energy from the sun to usable electric power.
Metal chalcogenide perovskites, with their nontoxic elemental composition, are known to offer greater thermal and aqueous stability than organic-inorganic halide perovskites. This means that they may be more suitable than other materials in the perovskite family to address the two biggest issues in commercial solar cell production: low thermal stability and toxicity.
Pagination
- Previous page
- Page 2
- Next page