Perovskites are materials that share a crystal structure similar to the mineral called perovskite, which consists of calcium titanium oxide (CaTiO3).
Depending on which atoms/molecules are used in the structure, perovskites can possess an impressive array of interesting properties including superconductivity, ferroelectricity, charge ordering, spin dependent transport and much more. Perovskites therefore hold exciting opportunities for physicists, chemists and material scientists.
Lead-based perovskites have become key materials in photovoltaics research thanks to their facile solution processability and impressive performance. However, despite their great potential, a persistent threat on future use and commercialization is the issue of toxicity and lead-content.
Lead (and its oxide form) is highly hazardous to animals and humans. Hence, succeeding to develop lead-free perovskites that could be used instead of the lead-based ones is a much desired accomplishment. While producing lead-free perovskite materials is not very hard to do, and various candidates exist (Sn-based perovskites, for example) these tend to perform poorly compared to lead-based perovskite materials, and exhibit limited optoelectronic performance and stability. However, vigorous research is taking place and hopefully it will yield lead-free perovskites that can serve as a viable replacement to lead-based ones.
The latest Perovskite toxicity news:
Research team advanced toward nontoxic perovskite solar cells
A team of scientists at Washington University in St. Louis has found what may be a more stable, less toxic semiconductor for solar applications using a novel double perovskite oxide, discovered through data analytics and quantum-mechanical calculations.
 An atomic model of KBaTeBiO6 (left), scanning transmission electron micrograph showing the atomic structure of KBaTeBiO6, along with snapshot of the synthesized powder (right). Credit: WUSTL
An atomic model of KBaTeBiO6 (left), scanning transmission electron micrograph showing the atomic structure of KBaTeBiO6, along with snapshot of the synthesized powder (right). Credit: WUSTL
Rohan Mishra, assistant professor of mechanical engineering & materials science in the McKelvey School of Engineering, led an interdisciplinary, international team that discovered the new semiconductor, made up of potassium, barium, tellurium, bismuth and oxygen (KBaTeBiO6). The lead-free double perovskite oxide was one of an initial 30,000 potential bismuth-based oxides. Of those 30,000, only about 25 were known compounds.
KAIST team proposes lead-free, efficient perovskite material for photovoltaic cells
A KAIST research team has proposed a perovskite material, Cs2Au2I6 that serves as a potential active material for highly efficient lead-free thin-film photovoltaic devices. This material is expected to lay the foundation to overcome previously known limitations of perovskite including its stability and toxicity issues.
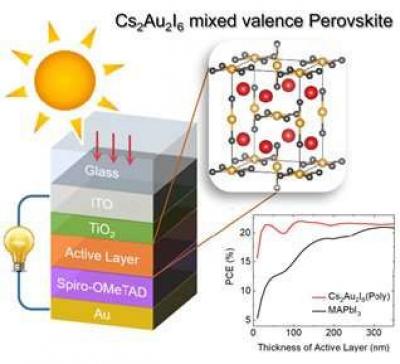
The joint team led by Professor Hyungjun Kim from the KAIST Department of Chemistry and Professor Min Seok Jang from the School of Electrical Engineering analyzed a previously discovered perovskite material, Cs2Au2I6, consisting of only inorganic substances and investigated its suitability for application in thin-film photovoltaic devices. Theoretical investigations suggests that this new perovskite material is not only as efficient but also more stable and environment friendly compared to the conventional perovskite materials.
Progress towards lead-free perovskite solar cells
Researchers from Sweden's Linköping University and the Nanyang Technological University (NTU) in Singapore have developed high-quality films based on double perovskites, which demonstrate promising photovoltaic properties. Developing environmentally friendly perovskites has become important in solving the toxicity issue of lead'based perovskite solar cells.
 The lead-free double perovskite solar cells (yellow, in the front) compared with the lead-based device (dark, in the background)
The lead-free double perovskite solar cells (yellow, in the front) compared with the lead-based device (dark, in the background)
"Our colleagues at Nanyang Technological University in Singapore have shown that the charge carriers demonstrate long diffusion lengths in the material, which is necessary if the material is to be appropriate for the application of in solar cells," says the Linköping team.
Chinese scientists synthesize lead-free double perovskite nanocrystals
A research team at the Dalian Institute of Chemical Physics (DICP) in the Chinese Academy of Sciences synthesized a new lead-free double perovskite nanocrystals (NCs) and revealed the hot-carrier dynamic of it.
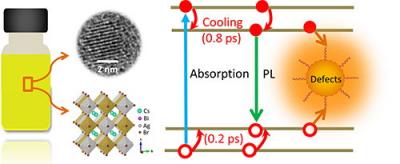
To avoid the toxicity issue of Pb, many efforts of finding a possible replacement are made. "We prepared the lead-free 3D double perovskite NCs and demonstrated that the continuously tunable emission ranged from 395 to 575 nm," said the researchers
New titanium-based material shows promise for lead-free perovskite-based PV
Researchers at Brown University and University of Nebraska - Lincoln (UNL) have come up with a new titanium-based material for making lead-free, inorganic perovskite solar cells. The team shows that the material has significant potential, especially for making tandem solar cells.
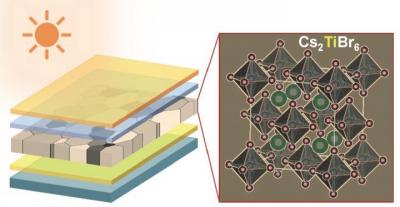
"Titanium is an abundant, robust and biocompatible element that, until now, has been largely overlooked in perovskite research," said the senior author of the new paper. "We showed that it's possible to use titanium-based material to make thin-film perovskites and that the material has favorable properties for solar applications which can be tuned."
Unique tin-based perovskite solar cells sport slow 'hot electrons' that can improve solar cell efficiency
Researchers at the University of Groningen are working on a special type of solar cell that is made of organic-inorganic hybrid perovskites. The team has been focusing on a material in which hot electrons retain their high energy levels for much longer, which might make it possible to use more of their energy to obtain a higher voltage.
Most hybrid-perovskite solar cells contain lead, which is toxic. The research group recently published a paper describing 9% efficiency in a hybrid-perovskite solar cell containing tin instead of lead. "When we studied this material further, we observed something strange", the team said. The results showed that the hot electrons produced in the tin-based solar cells took about a thousand times longer than usual to dissipate their excess energy.
Warwick U team designs tin-based perovskite solar cells
Researchers at the University of Warwick in the UK designed an environmentally friendly perovskite solar cell in which lead is substituted for tin with reportedly undiminished rates of performance but at cheaper cost and with lower toxicity.
The team stated that tin-based perovskites are much more stable than previously thought, and also render solar power cheaper, safer and possibly even more commercially attractive. "The device structure can be greatly simplified without compromising performance, which leads to the important advantage of reduced fabrication cost" the scientists say.
Oxford team realizes reduced toxicity solvent for 'greener' perovskite solar cells
Scientists at Oxford University have developed a solvent system with reduced toxicity that can be used to manufacture perovskite solar cells, which may clear one of the barriers to the commercialization of a technology.
By combining methylamine and acetonitrile, researchers have developed a clean solvent with a low boiling point and low viscosity that quickly crystallises perovskite films at room temperature and could be used to help coat large solar panels with the material.
Pagination
- Previous page
- Page 3