Researchers use perovskites to develop “multielement ink” – a “high-entropy” semiconductor that can be processed at low-temperature or room temperature
Researchers from Lawrence Berkeley National Laboratory (Berkeley Lab) and UC Berkeley have developed room-temperature-solution (20 °C) and low-temperature-solution (80 °C) synthesis procedures for a new class of metal halide perovskite high-entropy semiconductor (HES) single crystals. The “multielement ink” could enable cost-effective and energy-efficient semiconductor manufacturing and accelerate the sustainable production of next-gen microelectronics, photovoltaics, solid state lighting, and display devices.
“The traditional way of making semiconductor devices is energy-intensive and one of the major sources of carbon emissions,” said Peidong Yang, the senior author on the study, a faculty senior scientist in Berkeley Lab’s Materials Sciences Division and professor of chemistry and materials science and engineering at UC Berkeley. “Our new method of making semiconductors could pave the way for a more sustainable semiconductor industry.”
Researchers report self-assembled monolayer–based blue perovskite LEDs
Researchers from the University of Toronto, Peking University and Soochow University have studied the origins of unwanted emission in monolayer perovskite LEDs when the active layer thickness approaches ~5 nm and found that using available fabrication techniques results in a rough perovskite/HTL interface which leads to punch-through and direct electrical interaction between HTL and ETL (electron-transporting layer), and consequently, to undesired exciplex emission in LEDs.
The team sought to control monolayer interfaces in Rec.2100 primary blue perovskite LEDs and recognized that a well-defined, ordered, and compact monolayer film could suppress HTL/ETL interaction. They reasoned that this could be achieved if they could alter the polarity of the CsPbBr3 c-NC surface and thereby induce perovskite self-assembly down to the monolayer limit [i.e., self-assembled monolayer (SAM)] through the use of an HTL-compatible ligand. Self-assembled films with ordered nanocrystal arrangement maximize the interactions between nanocrystals and provide homogeneity needed for monolayer films with ~5-nm thickness.
Researchers design novel quantum random number generation based on a perovskite light emitting diode
Researchers from Linköping University in Sweden and Universidad de Concepción in Chile recently designed a new type of random number generator for encryption, based on Perovskiye LEDs. The new technology could make digital information exchange safer, cheaper and more environmentally friendly and even pave the way for a new type of quantum communication.
To encrypt information, a random number generator is used, which can either be a computer program or the hardware itself. The random number generator provides keys that are used to both encrypt and unlock the information at the receiving end. Different types of random number generators provide different levels of randomness and thus security. Hardware is the safer option as randomness is controlled by physical processes. And the hardware method that provides the best randomness is based on quantum phenomena – what researchers call the Quantum Random Number Generator, QRNG.
Researchers develop ballpoint pens that can write perovskite LEDs on diverse substrates
Researchers from Washington University in St. Louis and Florida State University have developed a versatile, scalable and eco-friendly handwriting approach to draw multicolor perovskite light-emitting diodes and perovskite photodetectors on various substrates, including paper, textiles, plastics, elastomers, rubber and three-dimensional objects.
The team's method uses common ballpoint pens filled with newly formulated inks of conductive polymers, metal nanowires and multiple perovskites for a wide range of emission colors. Just like writing with multicolored pens, writing layer-by-layer with these functional inks enables perovskite optoelectronic devices to be realized within minutes.
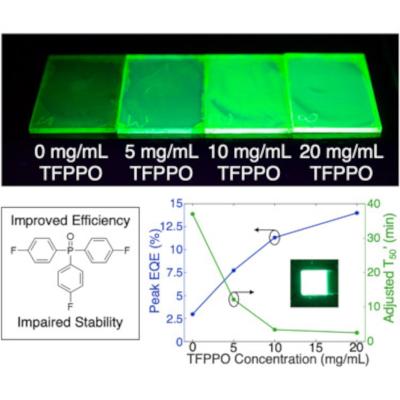
Researchers explore the effects of a molecular additive for perovskite LEDs
Researchers from Stanford University and Mississippi State University recently explored the potential of Mn2+-doped perovskite LEDs (PeLEDs) for lighting and display applications.
By introducing a molecular additive, tris(4-fluorphenyl)phosphine oxide (TFPPO), Mn2+-doped PeLEDs achieved a peak external quantum efficiency of 14.0% and peak luminance (i.e., brightness) of 128,000 cd/m2. These high efficiencies and brightnesses suggest that Mn2+-doped PeLEDs could be implemented in lighting or display applications. However, device stability is also important to consider. The team found that introducing TFPPO compromises the stability of Mn2+-doped PeLEDs—a decrease from 37.0 to 2.54 min. By analyzing both the optoelectronic and photophysical characteristics of Mn2+-doped PeLEDs before and after device operation, the scientists reported insights into this efficiency-stability trade-off.
Researchers use perovskites to design LEDs that reveal rot in food before it is visible
Researchers at the Indian Institute of Science Education and Research (IISER) have developed new LEDs which emit light simultaneously in two different wavelength ranges, for a simpler and more comprehensive way to monitor the freshness of fruit and vegetables.
The team explains that modifying the LEDs with perovskite materials causes them to emit in both the near-infrared range and the visible range, a significant development in the contact-free monitoring of food. Angshuman Nag and his team at the Indian Institute of Science Education and Research (IISER) are proposing a perovskite application in LED technology that could simplify the quality control of fresh fruit and vegetables.
Researchers develop perovskite LEDs with an external quantum efficiency exceeding 30%
Researchers from Xiamen University and China Jiliang University have reported ultrahigh-efficiency green PeLEDs with quantum efficiencies surpassing a milestone of 30% by regulating the charge carrier transport and near-field light distribution to reduce electron leakage and achieve a high light outcoupling efficiency of 41.82%.
The scientists applies Ni0.9Mg0.1Ox films with a high refractive index and increased hole carrier mobility as the hole injection layer to balance the charge carrier injection and insert the polyethylene glycol layer between the hole transport layer and the perovskite emissive layer to block the electron leakage and reduce the photon loss.
Stanford team shows how water additives improve the efficiency of violet perovskite light-emitting diodes
Researchers from Stanford University have reported a novel way to improve the performance of perovskite light-emitting diodes (PeLEDs) in the violet region. The new method could represent a promising step toward cost-effective and efficient ultraviolet PeLEDs.
By engineering the crystallization process of spin-coated 2D perovskites with water, the resulting PeLEDs exhibit bright violet emission at 408 nm with an external quantum efficiency of 0.41%, a 5-fold increase over control devices.
Researchers design ITO-free silicon-integrated perovskite electrochemical cell for light-emission and light-detection
Researchers from Russia's Alferov University, ITMO University, Far Eastern Branch of Russian Academy of Sciences, Peter the Great St. Petersburg Polytechnic University, Skolkovo Institute of Science and Technology and China's Qingdao Innovation and Development Center have developed a novel design for a perovskite electrochemical cell for light-emission and light-detection, where the active layer consists of a composite material made of halide perovskite microcrystals, polymer support matrix, and added mobile ions.
Schematic diagrams of (a) the typical PeLED device structure, where CTL - charge transfer layer, QD - quantum dots and (b) the team's PeLEC device structure, where SWCNT - single-walled carbon nanotubes. Image from Opto-Electronic Advances.
The team explained that while halide perovskite light-emitting devices exhibit exceptional properties such as high efficiency, high color purity, and broad color gamut, their industrial integration generally suffers from the technological complexity of devices' multilayer structure alongside in-operation induced heating poor stability. Halide perovskite light-emitting electrochemical cells are a novel type of perovskite optoelectronic device that differs from the perovskite light-emitting diodes by a simple monolayered architecture.
Perovskite-Info launches a new edition of its Perovskite for Displays Market Report
Perovskite-Info is proud to announce an update to our Perovskite for the Display Industry Market Report. This market report, brought to you by the world's leading perovskite and OLED industry experts, is a comprehensive guide to next-generation perovskite-based solutions for the display industry that enable efficient, low cost and high-quality display devices. The report is now updated to May 2023, with all the latest commercial and research activity.
Reading this report, you'll learn all about:
- Perovskite materials and their properties
- Perovskite applications in the display industry
- Perovskite QDs for color conversion
- Prominent perovskite display related research activities
The report also provides a list of perovskite display companies, datasheets and brochures of pQD film solutions, an introduction to perovskite materials and processes, an introduction to emerging display technologies and more.
Pagination
- Previous page
- Page 3
- Next page