Researchers from the Indian Institute of Science Education and Research Thiruvananthapuram (IISER-TVM), Germany's University of Potsdam and The Chinese University of Hong Kong have addressed an important aspect in the field of perovskite solar cells (PSCs) – the exact role of excess lead iodide content within the perovskite layer. While an optimal amount of excess lead iodide contributes to improved grain boundary passivation and blocking of minority charge carriers, leading to the development of highly efficient PSCs, the photo-stability of PSCs with surplus lead iodide remains a major concern. This concern stems from the catalytic role excess lead iodide can play in the degradation of PSCs under illumination.
The issue often arises during the fabrication of perovskite films using a two-step spin coating method, where the conversion of lead iodide films to perovskite is hindered due to challenges in controlling the reaction between lead iodide films and cationic precursor solutions. Various modifications of the two-step approach are presented in the literature, each aiming to achieve a near full conversion of lead iodide films into perovskite when exposed to cationic precursor solutions.
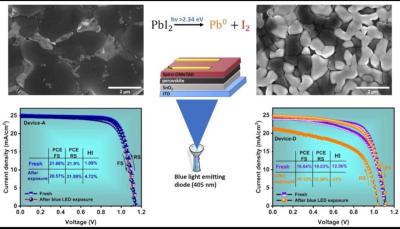
In their recent work, the team performed a comprehensive investigation on the evolution of excess lead iodide within four distinct variations of the two-step spin coating process. Notably, the power conversion efficiencies of all four versions were remarkably high, with the best efficiency being 23.4%.
Their research gives emphasis to the pivotal role processing methods play in formation of residual lead iodide within perovskite films, subsequently influencing the photovoltaic performance and stability of the resulting PSCs.
Additionally, the team systematically explored lead iodide degradation upon illumination, a factor that adversely impacts PSC hysteresis and stability. The scientists attributed the observed high hysteresis post-illumination to the decomposition of lead iodide into metallic lead and iodine, a conclusion corroborated by x-ray photoelectron spectroscopy findings. These insights align with the calculated ion-migration activation energy.