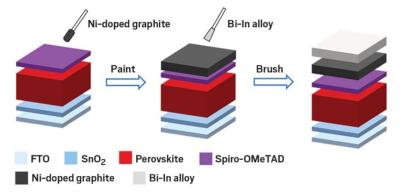
Researchers from Northern Illinois University, National Renewable Energy Laboratory (NREL), Northwestern University and Argonne National Laboratory have reported a bilayer back electrode configuration consisting of an Ni-doped natural graphite layer with a fusible Bi-In alloy. This back electrode can be deposited in a vacuum-free approach and enables perovskite solar cells (PSCs) with a power conversion efficiency of 21.0%. These inexpensive materials and facile ambient fabrication techniques can help provide an appealing solution to low-cost PSC industrialization.
A thin layer of gold or silver can help improve the efficiency of perovskite solar cells, but the researchers have found a less expensive material that will enable commercialization of the technology without exorbitant cost. “A layer of gold in a solar panel or even a layer of silver is probably too expensive,” said Kai Zhu, a senior scientist in the Chemistry and Nanoscience Center at the U.S. Department of Energy’s National Renewable Energy Laboratory (NREL). “It would make the solar panel not affordable for most people.”
Instead of a precious metal, Zhu and his colleagues have a another, cheaper idea. The solution is a nickel-doped graphite layer coupled with a bismuth-indium alloy layer. The two layers can be easily integrated into the perovskite device through painting them on, offering a low-cost fabrication method.
“Our team has identified a potentially disruptive technology that could help reduce the infrastructure investment for use of highly promising perovskite solar cells in solar panels,” said Tao Xu, a chemistry professor at Northern Illinois University and Zhu’s co-corresponding author of the recent paper. “Our approach replaces costly gold, commonly used to make the back-metal electrode in these solar cells through an expensive high-temperature vacuum-chamber process. Instead of gold, we use inexpensive materials that can be readily laminated to thin films at atmospheric pressure and mild temperatures. We think this will be an appealing low-cost solution that could help speed commercialization of perovskite solar cells.”
Zhu said the perovskite solar cell made with the new materials yielded a laboratory efficiency of 21%. Further research should boost the efficiency higher and closer to that of perovskites made with precious metals, which hold a record efficiency of 26%. Metal is better than carbon at conducting electricity.
Perovskite solar cells are made by depositing chemicals onto a substrate. Individual layers all serve a purpose, with the perovskite layer serving as the semiconductor. The photons from sunlight trigger electrons to move in one direction, creating a vacancy—or “hole”—that then moves in the opposite direction. The right energy level is needed to spark this movement, in the process creating an electric current. The graphite material and gold both possess the right energy level.
By eliminating a layer of precious metals, the cost to manufacturer perovskites will be significantly cheaper, the researchers calculated. An analysis suggests that for a perovskite-based solar plant with a gigawatt of power output, using the graphite/alloy bilayer will slash costs of the contact electrodes by a factor of between 4 and 1,000 depending on the other types of materials used for back materials.
“That’s the selling point for this approach,” Zhu said.