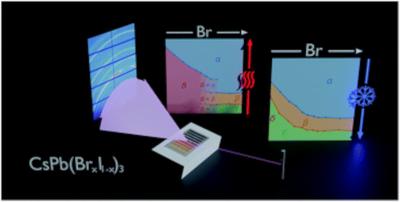
Researchers at HZB have reported findings from their recent work: printing and exploring different compositions of caesium based halide perovskites (CsPb(BrxI1'x)3 (0 ⤠x ⤠1)).
In a temperature range between room temperature and 300 Celsius, the team observed structural phase transitions influencing the electronic properties. The study presents a quick and easy method to assess new compositions of perovskite materials in order to identify potential candidates for applications in thin film solar cells and optoelectronic devices.
Now, the HZB team explored compositions of CsPb(BrxI1'x)3, which provide tunable optical band gaps between 1.73 and 2.37 eV. This makes these mixtures really interesting for multi-junction solar cell applications, in particular for tandem devices.
For the production process, the researchers used a newly developed method for printing combinatorial perovskite thin films to produce systematic variations of CsPb(BrxI1'x)3 thin films onto a substrate. To achieve this, two print heads were filled with either CsPbBr2I or CsPbI3 and then programmed to print the required amount of liquid droplets onto the substrate to form a thin film of the wanted composition. After annealing at 100 Celsius to drive out the solvent and crystallize the sample, they obtained thin stripes with different compositions.
With a special high intensity x-ray source, the liquid metal jet in the LIMAX lab at HZB, the crystalline structure of the thin film was analyzed at different temperatures, ranging from room temperature up to 300 Celsius. "We find that all investigated compositions convert to a cubic perovskite phase at high temperature," Hampus Näsström, PhD student and first author of the publication explains. Upon cooling down, all samples transition to metastable tetragonal and orthorhombic distorted perovskite phases, which make them suitable for solar cell devices. "This has proven to be an ideal use case of in-situ XRD with the lab-based high-brilliance X-ray source," Roland Mainz, head of the LIMAX laboratory, adds.
Since the transition temperatures into the desired phases are found to decrease with increasing bromide content, this would allow to lower processing temperatures for inorganic perovskite solar cells.
"The interest in this new class of solar materials is huge, and the possible compositional variations near to infinite. This work demonstrates how to produce and assess systematically a wide range of compositions," says Dr. Eva Unger, who heads the Young Investigator Group Hybrid Materials Formation and Scaling. Dr. Thomas Unold, head of the Combinatorial Energy Materials Research group agrees and suggests that "this is a prime example of how high-throughput approaches in research could vastly accelerate discovery and optimization of materials in future research."

